In June 2023, we commenced a project to review and update the Maternity and Neonatal eHandbook guidelines, with a view to targeting completion in 2024. Please be aware that pending this review, some of the current guidelines may be out of date. In the meantime, we recommend that you also refer to more contemporaneous evidence.
Women who are obese have increased risks of pregnancy complications and their babies are at risk of poorer outcomes (Table 1).
There has recently been increased attention on optimal management of women who are pregnant and obese. However, few recommendations are based on Level I evidence. This guidance synthesises current best practice and provides resources for further reading.
Table 1: Risks and problems associated with BMI >30 in pregnancy
| Maternal | Anaesthetic | Fetal and neonatal |
|---|---|---|
|
|
|
Body mass index (BMI)
BMI is defined as weight in kilograms divided by the square of the height in metres (kg/m2). See below.
Although there are some concerns about the reliability of BMI in pregnancy, it is a practical tool to determine relevant risk of complications.
It is recommended that:
- BMI be based on height and weight measured in early pregnancy (ideally less than 12 weeks)
- measuring and documenting height and weight form part of the initial clinical assessment of pregnancy of every woman, either in the community or at first hospital visit, whichever is earliest.
Classification of BMI
The World Health Organization (WHO) classifies obesity according to BMI (Table 2).
Most pregnancy complications associated with obesity are related to the degree of obesity at booking.
Table 2: WHO Classification of obesity according to BMI
| Classification | BMI (kg/m2) | Risk of co-morbidities |
|---|---|---|
| Underweight | Less than 18.5 | Low (but risk of other clinical problems increased) |
| Normal range | 18.5–24.9 | Average |
| Overweight | 25–29.9 | Increased |
| Obese I | 30–34.9 | Moderate |
| Obese II | 35–39.9 | Severe |
| Obese III | Greater than or equal to 40.0 | Very severe / extreme |
Facility capabilities
- Health services should provide planned care to pregnant women based on a risk management approach that establishes when advice, referral, or transfer of obese women is required. This should align with the Capability Framework for Victorian Maternity and Newborn Services.
- Services that determine a cut-off BMI due to physical and service delivery capabilities should develop a management plan outlining their response to unplanned admissions of women above the cut-off.
- Assessment of physical and service delivery capabilities should include (but not be limited to):
- capability to manage the potential risks and complications of obesity
- workforce capabilities, for example limitations to access to a range and number of appropriately skilled healthcare professionals
- facility design, such as width of access doors and pathways, turning circles for large patient equipment
- availability of large patient equipment with appropriate safe working loads and widths.
Referral and transfer
- Most relative risk data are based on early pregnancy or pre-pregnancy BMI.
- Plan for transfer early: transfer of care to a service with appropriate facilities for safe care of the woman and her baby must occur early in the care and must include consistent information sharing with women.
- Consider options for pregnancy and postpartum care, in addition to the safest place for the birth. For example, shared care between the local and higher-level service may enable pregnancy and postpartum care to be provided close to the woman’s home.
- Implications of late pregnancy BMI are less clear. If a woman has gained weight and reaches above the threshold for certain equipment, late referral might be recommended.
- Consider each individual: consider referral for women close to the BMI transfer threshold, especially if they have gained significant weight in pregnancy or the BMI was calculated after 20 weeks.
- More information to inform individualised care planning:
- It is strongly recommended that obese pregnant women are not booked for a planned home birth.
Transport capacity
Emergency transfer of women of high BMI by Ambulance Victoria (AV), or other emergency transport such as the Royal Flying Doctor Service, is limited by restrictions in the weight capacity of vehicles and, in some cases, the overall dimensions of the woman (Table 3). These restrictions underscore the importance of early, planned transfer of care.
Table 3: Transport weight restrictions for Ambulance Victoria
| Non-emergency stretcher | Standard Stretcher | Complex patient ambulance vehicle | Rotary Wing | Fixed Wing | |
|---|---|---|---|---|---|
| Patient weight limit (Safe Working Load) | 159kg | 230kg | 318 kg lifting capacity with no lift assistance 454 kg transport capability in lowest position | 140–160kg | 120kg |
Antenatal care
Multidisciplinary care
Care of an obese pregnant woman requires input from midwives, obstetricians, general practitioners, ultrasonographers, anaesthetists, dietitians, endocrinologists, haematologists, obstetric medicine specialists, physiotherapists and other allied health clinicians, depending on her individual risks and comorbidities.
Referral for consultation at a higher-level service may be required where local access is limited.
A ‘low risk’ schedule or approach to antenatal care is not appropriate.
Practice points
- Inform women of their increased risk of neural tube defects.
- Advise all women to take high dose (5 mg) folic acid supplementation during the first trimester.
- Reinforce the need to monitor and respond to decreased fetal movements.
- Be aware of the increased risks of pre-eclampsia and fetal growth restriction: consider low dose aspirin (150 mg nocte) from <16 weeks.
- Remember thromboprophylaxis.
- Be aware of the increased risk of peripartum depression: PANDA Anxiety and Depression in Pregnancy.
- Advise women to stop taking prescription and over-the-counter weight loss medications and complementary alternative medications.
- Recommend more frequent antenatal visits in the third trimester to monitor for complications: fortnightly visits from 28 weeks and weekly visits from 36 weeks is suggested.
- In the third trimester, consider an individual mobility assessment to identify equipment, workforce and procedural requirements for safe delivery of care.
Counselling, nutrition and physical activity
Conversations about obesity can be challenging for women and care providers, and information about associated perinatal risks can be confronting. Care providers should be aware of the potential for women to experience feelings of stigma and shame, and counselling must be sensitive and non-judgmental.
- Discuss risks of obesity early in pregnancy.
- Explain the woman’s individual antenatal care schedule and describe its importance in monitoring for complications.
- Discuss family diet and eating patterns, for example:
- advise women to eat a healthy diet as per the Australian Guide to Healthy Eating
- advise women not to restrict dietary intake below the recommended food group requirements for pregnancy
- consider referral to a dietitian.
- Identify physical activity goals (table 4):
- in the absence of obstetric or medical complications, advise women to undertake 30–60 minutes of moderate activity at least 3–4 times per week (tables 4 and 5).
- consider physiotherapy consultation to assist with assessment and individual exercise prescription.
- Help women and their partners identify supports to help them meet their goals.
Table 4. Safe and effective exercise in pregnancy
| When to start | First trimester <12 weeks gestation |
|---|---|
| Duration of a session | 30–60 minutes |
| Times per week | At least 3–4 (up to daily) |
| Intensity of exercise | <60–80% of age-predicted maximum maternal heart rate1 |
| Self-reported intensity of exercise | Moderate intensity (12–14 on Borg scale2) |
| Supervision of exercise | Preferred, if available |
| When to end | Until delivery (as tolerated) |
1Usually not exceeding 140 beats per minute.
2Borg scale is a 15-category scale (from 6-20) to measure the level of perceived exertion: light exercise is approximately 6-11; 13 is somewhat hard; 15 is hard; 19 is extremely hard.
Berghella. Exercise in pregnancy! Am J Obstet Gynecol 2017.
Table 5. Types of beneficial exercises in pregnancy
| Walking |
| Aerobic exercises |
| Stationary cycling |
| Stretching exercises |
| Dancing |
| Hydrotherapy, water aerobics |
| Resistance exercises (e.g. weights, elastic bands) |
This is a partial list - the full list of all safe and studied exercises in pregnancy would be too long to publish.
Berghella. Exercise in pregnancy! Am J Obstet Gynecol 2017.
Gestational weight gain
- Excessive gestational weight gain has been correlated with:
- fetal macrosomia
- operative vaginal delivery
- caesarean section
- low Apgar scores
- admission to neonatal intensive care.
- Help women and their partners understand that limiting gestational weight gain can reduce the risk of adverse outcomes.
- Routinely discuss appropriate weight gain and:
- support women to identify gestational weight gain goals (table 6)
- advise women that limited weight gain, rather than weight loss, is the primary goal of weight management during pregnancy.
- More restrictive weight gain and possibly no weight gain may be appropriate for women with extreme obesity (BMI >50).
- Weight loss programs are not appropriate for most pregnant women.
- Recommended total weight gain for an obese woman pregnant with twins is 11–19 kg.
Table 6. Gestational weight gain goal ranges
| Pre-pregnancy BMI | Rate of gain 2nd and 3rd trimester (kg/week)* | Recommended total gain range (kg) |
|---|---|---|
| Less than 18.5 | 0.45 | 12.5 to 18 |
| 18.5 to 24.9 | 0.45 | 11.5 to 16 |
| 25.0 to 29.0 | 0.28 | 6.8 to 11.3 |
| Greater than or equal to 30.0 | 0.22 | 5 to 9.1 |
Pregnancy after bariatric surgery
It is now common to care for women with a history of bariatric surgery.
Practice points
- Seek input, where possible, from the woman’s bariatric surgeon and dietitian.
- Document the type and date of bariatric surgery in the woman’s pregnancy record: the first two years post-surgery is the period of maximal weight loss and associated risk of fetal growth restriction.
- Monitor for fetal growth restriction: ultrasound scan (US) for growth at 28, 32 and 36 weeks.
- Encourage continuation of prescribed nutritional supplements (in particular B12, folate and iron).
- Evaluate for nutritional deficiencies and correct with appropriate supplements as indicated.
- Check ferritin, vitamin B12, folate, calcium and vitamin D in each trimester and treat as indicated.
- Maintain a high index of suspicion and high level of clinical surveillance for intestinal obstruction: this may present as abdominal complaints, morning sickness, reflux or uterine contractions.
- Early pregnancy nausea and vomiting may require partial or complete deflation of laparoscopic–adjustable gastric banding.
- Bariatric surgery is not a contraindication to breastfeeding. Maternal nutritional supplementation may be required.
Maternal investigations
Increased screening for potential comorbidities and pregnancy complications may help prevent late diagnosis and aid timely intervention.
Symphyseal fundal height (SFH) measurement:
- BMI 30-<40: measure SFH at every visit from 24 weeks
- If SFH measurements are reduced, or show slow or static growth on serial measurements, refer the woman for US assessment of fetal growth
- BMI >/= 40: use serial US assessment to monitor fetal growth.
Ultrasound assessment
- Document the woman’s current BMI on all US referrals.
- Conduct a first trimester US for accurate dating, diagnosis of twins or higher order multiples and increased accuracy of first trimester screening.
- Assess appropriateness of growth parameters in the context of BMI: US assessment of growth is hindered in obese women.
- US must be undertaken and interpreted by a clinician skilled and experienced in scanning obese women.
- Perform anomaly scanning at 20–23 weeks.
- A growth scan is recommended at 35 weeks for women with a BMI of ≥35 and <40.
- Serial assessment of growth at 28, 32 and 36 weeks is recommended for women with a BMI ≥40.
Blood tests
- Refer all women for a 75 g Oral Glucose Tolerance Test (OGTT) at 14-16 weeks.
- Repeat OGTT at 26–28 weeks if initial test is negative.
- BMI 30–<40: consider baseline investigations of renal function in early pregnancy, to assist in diagnosis and management later in pregnancy.
- BMI ≥40: test for baseline renal function (presence of proteinuria, serum creatinine and urea) and liver function. This will help distinguish chronic renal dysfunction secondary to maternal chronic hypertension and/or diabetes from pregnancy associated hypertensive disorders.
Assessment of comorbidities
- Measure and document upper arm circumference: ensure appropriate cuff size when assessing blood pressure.
- Be aware of the increased risks of pre-eclampsia and fetal growth restriction: consider low dose aspirin (150 mg nocte) from <16 weeks.
- Remember thromboprophylaxis.
- Consider cardiac risk assessment for women with pre-existing medical conditions: particularly for women with a BMI ≥40 with additional risk factors, such as smoking and type 2 diabetes.
- Obesity is one of the strongest risk factors for obstructive sleep apnoea, which may worsen during pregnancy:
- BMI ≥40: assess in early pregnancy using the STOP Questionnaire (table 7) to facilitate timely management.
Table 7. STOP Questionnaire
| S | Do you snore loudly (louder than talking or loud enough to be heard through closed doors)? |
| T | Do you often feel tired, fatigued or sleepy during day time? |
| O | Has anyone observed you stop breathing during your sleep? |
| P | Do you have or are you being treated for high blood pressure? |
If the answer is yes to 2 or more questions, refer to a physician/sleep specialist*.
See: Australasian Sleep Association for more information.
Anaesthetic considerations
Up to 75 per cent of all anaesthesia-related maternal deaths occur in obese pregnant women. An anaesthetic review in pregnancy provides an opportunity to assess comorbidities, particularly those that increase anaesthetic risk, including difficulty of intubation, regional anaesthesia and obtaining venous access.
This is particularly pertinent to services without registered medical specialist - Australian and New Zealand College of Anaesthetists (ANZCA) - availability 24 hours a day and for services that may need to consider transfer on the basis of a woman’s BMI and/or comorbidities.
- Anaesthetic review recommendations:
- BMI ≥35 & Level 2/3 Service: refer all women for anaesthetic review
- BMI ≥35 & Level 4/5 Service: refer women with comorbidities for anaesthetic review
- BMI ≥40: refer all women for anaesthetic review
- develop and document an anaesthetic management plan in consultation with the multidisciplinary team.
Timing of delivery
- Risk of late pregnancy stillbirth is significantly higher in obese women: some studies show a risk of stillbirth for class III obesity (BMI ≥40) more than 10 times greater than the risk for healthy women (Figure 1).
- Induction of labour (IOL) may be appropriate earlier for obese women than for women with a healthy weight.
- After 39 weeks, counsel all women about the comparative risks and benefits of ongoing pregnancy versus induction around their due date.
- Benefit of induction is likely to be greatest for women with the highest levels of obesity: risk of stillbirth rises rapidly after 38 weeks for women with a BMI ≥50
- Optimal timing of delivery for obese women is also influenced by the presence of comorbidities, such as diabetes or pre-eclampsia.
Delivery is recommended at 38–39 weeks for women with a BMI ≥50.
Figure 1: Risk of stillbirth by gestational period
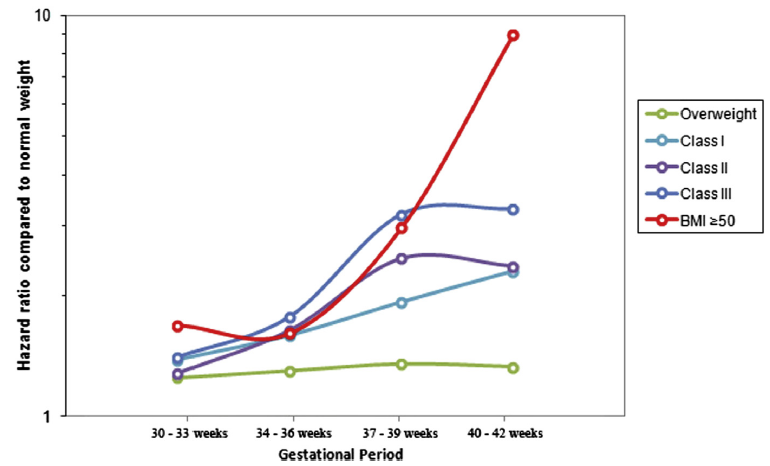 From: Yao. Obesity and the risk of stillbirth. Am J Obstet Gynecol 2014.
From: Yao. Obesity and the risk of stillbirth. Am J Obstet Gynecol 2014.
Induction of labour methods
- There is currently no clear evidence to favour one method of induction over another for obese women.
- Establishing active labour may take longer and obese women should be advised of the possibility of a protracted induction process.
- See - Induction of labour
Mode of birth
- There is a lack of evidence to guide mode of delivery advice.
- While there is an increased risk of caesarean birth for nulliparous women, overall maternal and neonatal morbidity does not appear to be reduced by elective caesarean: the decision should be based on individual assessment.
- There is an increase in complications and a reduction in success of VBAC relative to increasing BMI.
- The low likelihood of VBAC success, particularly for extremely obese women, the increased risk of uterine rupture and the difficulties in undertaking an emergency caesarean support a cautious approach to VBAC for these women.
Intrapartum care
- A number of obesity related risks increase the likelihood of intrapartum complications.
- Take a team approach, including frequent communication between care providers.
On admission
- Ensure appropriate equipment is accessible: bed, hoists, limb lifters, transfer equipment, US (may be required to visualise veins).
- Notify anaesthetic and theatre staff when obese women are admitted.
- Consider pre-siting an epidural for women having an IOL and requesting epidural anaesthesia.
- BMI ≥35: obtain intravenous access with at least 1 x 16 g IV cannula.
- BMI ≥40: obtain intravenous access with 2 x 16 g IV cannula.
First and second stage
- Place a hover mat under women having an epidural.
- Consider pressure area care for women with an epidural.
- Prepare for increased likelihood of shoulder dystocia.
- Prepare for potential need for blood and blood products in line with capability level requirements: Capability Framework for Victorian Maternity and Newborn Services.
- Continuous electronic fetal monitoring (CEFM) for women with a BMI ≥40.
- CEFM for women with a BMI >30 and other risk factors.
- If continuous fetal heart rate recording cannot be obtained externally, internal fetal monitoring should be commenced.
- Refer to local guidelines on water immersion for labour and birth.
- Consider potential need for neonatal resuscitation: ensure clinician/s with appropriate training attend birth.
Third stage
- Active management of third stage:
- women who request physiological management of the third stage should be advised of increased risks of postpartum haemorrhage (PPH) with obesity.
Caesarean section
The challenges of surgery on obese women, particularly women with a BMI ≥40, may be mitigated by adjustments to the operative approach (further reading).
Practice points
- Ensure surgeon and anaesthetist have the appropriate experience.
- Be aware of potential for difficult and failed tracheal intubation.
- Consider the option of a traction device for elevation of a pannus (for example, traxi, abdominal wall retractor).
- Consider increased prophylactic antibiotic dosing (for example, 3 g cephazolin IV) for women with a BMI ≥40.
- Administer antibiotics 60 minutes before skin incision.
- Consider vertical incision for some women.
- The subcutaneous layer should be closed.
- There is no benefit to subcutaneous drain placement.
- The benefit of one skin closure technique over another is unclear.
- Negative pressure wound therapy may be beneficial for women with a BMI ≥40.
Postpartum care
- Increased observation, including respiratory rate, may be required due to the risk of airway compromise, obstructive sleep apnoea and aspiration, particularly following administration of narcotic and sedative medications.
- Increased breastfeeding support may be required.
Immediate postpartum care
- Consider postpartum thromboprophylaxis:
- anticoagulant dosage should be appropriate for maternal weight
- graduated compression stockings and/or calf compression.
- If a woman has received sedatives or narcotics, keep the bed head at a 45 degree angle until she is alert.
- Encourage early mobilisation and regular physiotherapy, particularly of women who have had a caesarean birth.
- Remember the risk of pressure injuries during periods of immobility:
- perform and document skin inspection
- discuss the risk of pressure injury with the woman.
- Increase clinical surveillance for signs of infection, including regular wound care (abdominal and perineal) and thorough assessment of elevated maternal temperature.
Pre-discharge care
- Ensure communication and clinical handover with the woman’s Maternal and Child Health Nurse and GP, including advice on specific risk mitigation measures as determined by the pregnancy care plan.
- Encourage all women to continue with nutritional counselling and exercise programs postpartum.
- If hormonal methods of contraception are considered for use by the obese woman (particularly oestrogenic compounds), undertake a risk assessment of venous thromboembolism (VTE).
Follow up care
- Refer women with gestational diabetes for an OGTT at six weeks postpartum (Maternity eHandbook - Gestational diabetes).
Neonatal considerations
- Refer to the Neonatal ehandbook for care of the baby who is:
Thromboprophylaxis
- Obesity is an independent risk factor for VTE.
- Low molecular weight heparins (LMWH) such as enoxaparin and dalteparin are the drugs of choice for prevention and management of VTE in pregnancy and postpartum as they do not cross the placenta and are not excreted into breast milk.
- Consider increased dosing (0.5 mg/kg) for women over 120 kg.
Antenatal thromboprophylaxis
- Consider antenatal thromboprophylaxis from 28 weeks if there are three or more antenatal risk factors (Table 9).
Table 9. Antenatal VTE risk factors
| Age >35 years | Parity ≥3 |
|---|---|
| Smoker | Gross varicose veins |
| Pre-eclampsia | Immobility |
| Family history of unprovoked or oestrogen provoked VTE in a first degree relative | Low risk thrombophilia (Factor V Leiden or Prothrombin gene mutation heterozygosity) |
| Multiple pregnancy | IVF |
| Transient risk factors (hyperemesis, long haul travel, infection) |
Postpartum thromboprophylaxis
• Inpatient care: consider compression stockings and encourage early mobilisation.
• Consider postpartum thromboprophylaxis for 10 days if there is a single postpartum risk factor (Table 10).
• Consider postpartum thromboprophylaxis for six weeks if there are three or more postpartum risk factors (Table 10).
Table 10. Postpartum VTE risk factors
| Age >35 years | Parity ≥3 |
|---|---|
| Smoker | Caesarean section |
| Family history of VTE | Low risk thrombophilia |
| Gross varicose veins | Systemic infection |
| Immobility | Pre-eclampsia |
| Multiple pregnancy | Preterm delivery (12 hours) |
| Stillbirth | Prolonged labour (>12 hours) |
| PPH >1L |
Breastfeeding
Obese women are at an increased risk of delayed and unsuccessful lactation.
Practice points
- Consider antenatal referral to a lactation consultant.
- Ensure increased support and assistance during breastfeeding.
- Discourage co-sleeping.
- Offer early postpartum breastfeeding support.
- Provide information about breastfeeding support services (ABA, LSU services).
- Discuss the risks of SIDS/SUDI.
The risk of sudden unexplained death in infancy (SUDI) is significantly increased when a co-sleeping parent/s is/are obese.
Inter-pregnancy care
- Obesity is a modifiable risk factor: advise weight reduction as an important goal for women anticipating future pregnancies.
- An increase in pre-pregnancy weight between a first and second pregnancy from an overweight BMI to an obese BMI increases the risk of pre-eclampsia.
- Conversely, a decrease in pre-pregnancy weight between first and second pregnancies, from obese to a normal BMI, decreases the risk of caesarean and large-for-gestational age infants.
- Recommend high dose (5 mg/day) folic acid supplementation pre-conceptually for at least one month and throughout the first trimester.
Audit and performance improvement
All maternity services should have processes in place for:
- auditing clinical practice and outcomes
- providing feedback to clinicians on audit results
- addressing risks, if identified
- implementing change, if indicated.
Potential auditable standards include:
- antenatal recording of BMI
- appropriate gestational weight gain
- intrapartum IV cannulation as per standard of care
- women who have active management of third stage
- appropriate prescription of antenatal and postnatal thromboprophylaxis
- rates of women breastfeeding at discharge.
For more information or assistance with auditing, please contact the Maternity and Newborn Clinical Network: maternityehandbook@safercare.vic.gov.au
References
- Australian Commission on Safety and Quality in Health Care. Standard 5. Comprehensive care, minimising patient harm, 5.2 preventing and managing pressure injuries 2018
- Berghella. Exercise in pregnancy! Am J Obstet Gynecol 2017.
- Yao. Obesity and the risk of stillbirth. Am J Obstet Gynecol 2014.
Further reading
- Arden, Duxbury & Soltani (2014) What women really think about gestational weight management: A thematic analysis of posts made in online parenting forums. Pregnancy Hypertension: An International Journal of Women’s Cardiovascular Health. DOI: 10.1016/j.preghy.2014.03.007
- Ayres-de-Campos D. Obesity and the challenges of caesarean delivery: Prevention and management of wound complications. Best Practice & Research Clinical Obstetrics and Gynaecology 29 (2015) 406¬–414
- ACOG Practice Bulletin No. 105: Bariatric surgery and pregnancy: Obstet Gynecol. 2009; 113:1405-1413
- Capability Framework for Victorian Maternity and Newborn Services
- Cassidy, Copeland & Simpson (2014) Weight management in pregnancy: participants’ experiences of ‘Healthy Eating and Lifestyle in Pregnancy (HELP)’, a maternity care intervention for obese pregnant women. Pregnancy Hypertension: An International Journal of Women’s Cardiovascular Health. DOI: 10.1016/j.preghy.2014.03.012
- CMACE / RCOG Joint Guideline: Management of Women with Obesity in Pregnancy
- Consultative Council on Obstetric and Paediatric Mortality and Morbidity
- Keely, Cunningham-Burley, Elliott, Sandall & Whittaker (2016) “If she wants to eat…and eat and eat…fine! It’s gonna feed the baby”: Pregnant women and partners’ perceptions and experiences of pregnancy with a BMI >40kg/m2. Midwifery. DOI: 10.1016/j.midw.2016.09.016
- McParlin, Bell, Robson, Muirhead & Araujo-Soares (2016) What helps or hinders midwives to implement physical activity guidelines for obese pregnant women? A questionnaire survey using the Theoretical Domains Framework. Midwifery. DOI: 10.1016/j.midw.2016.09.015
- Queensland Statewide Maternity and Neonatal Clinical Guideline ‘Obesity’
- RANZCOG Management of obesity (2017)
Appendices
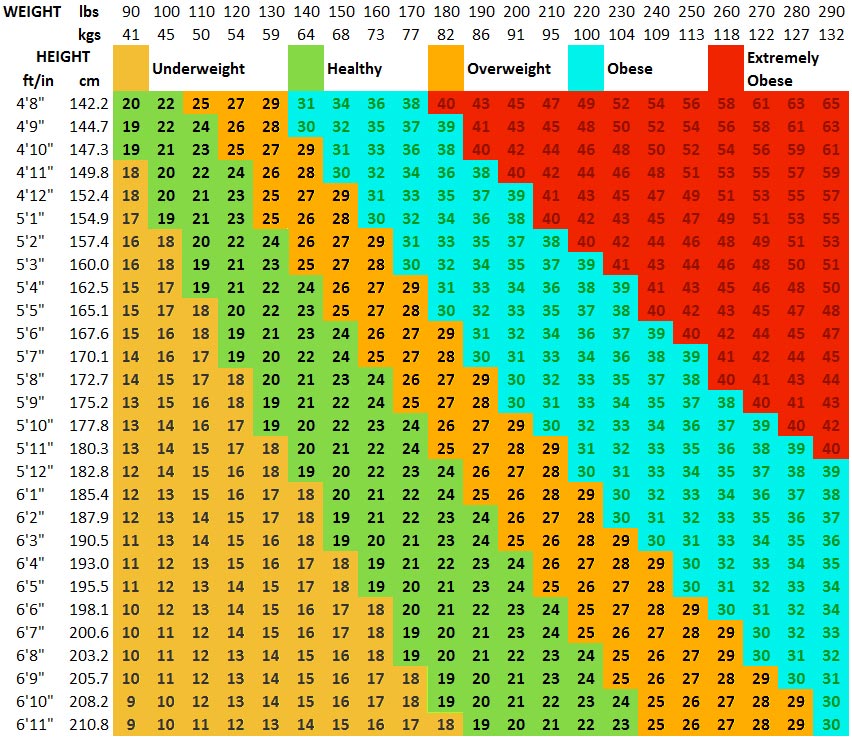 Figure 2: BMI chart
Figure 2: BMI chart
Abbreviations
| AV | Ambulance Victoria |
|---|---|
| ABA | Australian Breastfeeding Association |
| BMI | Body mass index |
| DFM | Decreased fetal movements |
| ICU/HDU | Intensive care unit/high dependency unit |
| IOL | Induction of labour |
| LSU | Lactation support unit |
| NIPT | Non-invasive pre-natal testing |
| OGTT | Oral glucose tolerance test |
| PPH | Postpartum haemorrhage |
| SIDS | Sudden Infant Death Syndrome |
| SUDI | Sudden Unexpected Death in Infancy |
Acknowledgements
This guideline was originally developed in 2011 by Glyn Teale, Debbie Rogers and Lisa Oro, management team of the Maternity and Newborn Clinical Network (the network) in consultation with members of the Obesity Guideline Working Group: Robert North, Euan Wallace, Michael Permezel, Denise Patterson, Cate Nagle, Serena Bridges and Scott Simmons. Contributions were also received from Anne Hotchin, Ines Rio, Kaye Gall, Kylie Osborne, Leonie Henderson, Michael Stewart, Tanya Farrell and Vanessa Watkins.
This updated version has been prepared by Glyn Teale and is endorsed by the Governance Group of the network. We acknowledge the contribution of Dr Briony Cutts to recommendations relating to thromboprophylaxis and Dr Samuel Mathew and Dr Jyoti Sharma to recommendations related to caesarean section in obese women.
We also acknowledge the Queensland Maternity and Neonatal Network, and the Centre for Maternal and Child Enquires / Royal College of Obstetricians and Gynaecologists (CMACE/RCOG) whose guidelines have been adapted in the development of the Victorian guideline.
Download
Get in touch
Version history
First published: August 2018
Due for review: November 2021

