Please note that some guidelines may be past their review date. The review process is currently paused. It is recommended that you also refer to more contemporaneous evidence.
Respiratory distress syndrome (RDS) is when the neonate has difficulty breathing due to surfactant deficiency at birth. RDS, also known as hyaline membrane disease (HMD), is the dominant clinical problem faced by preterm infants and is directly related to structurally immature and surfactant deficient lungs.
The greatest risk factor is low gestational age and the development of the disease begins with the impaired synthesis of pulmonary surfactant associated with prematurity.
The disease is exacerbated by treatable and preventable factors including:
- cold stress
- hypoxia
- acidosis
- secondary surfactant deficiencies - meconium aspiration syndrome, infant of diabetic mother, pneumonia.
The diagnosis is made on the basis of the combination of clinical features including:
- tachypnoea (generally > 60 breaths per minute in term and > 80 breaths per minute in preterm infants)
- nasal flaring
- grunting respirations
- intercostal retraction
- cyanosis
- increased oxygen requirement
radiological features.
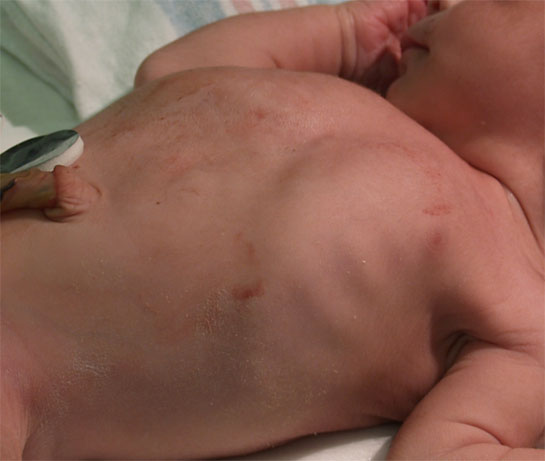 Figure 1: Intercostal retraction
Figure 1: Intercostal retraction
Photo courtesy of Janele Alby MD
The natural history is for the clinical signs to develop within six hours of life, with progressive worsening over the first 48-72 hours of life followed by recovery.
The condition can be prevented, or the severity reduced, by antenatal administration of betamethasone. The course of the disease is altered by exogenous surfactant therapy and assisted ventilation.
Attention to thermoregulation and oxygenation can decrease the severity of RDS.
The level of experience and expertise dictate what technical procedures (peripheral intravenous (IV) catheter insertion and ETT insertion) are used prior to arrival of the PIPER Neonatal transport team.
Infants requiring greater than 60 per cent oxygen should be managed in a Level 6 Neonatal Unit.
Differential diagnosis
Signs and radiolological appearance of RDS are not specific and other causes of respiratory distress should be considered.
In particular it is difficult to exclude sepsis as a possible diagnosis initially, and antibiotic therapy should be given until blood cultures prove negative.
‘Wet lung’ and lung malformations as well as non-pulmonary causes of respiratory distress are uncommon in the preterm infant but should be excluded using the appropriate tests.
Investigation
Chest x-ray
- This is useful and should be performed in all cases of respiratory distress once the oxygen requirement exceeds 30 per cent. The x-ray may need to be repeated if the illness runs a course that is unusual or protracted or the infant’s status suddenly worsens when air leak complications must be considered.
Chest radiograph
- typically gives diffuse ground glass lungs with low volumes and a bell-shaped thorax
- often tends to be bilateral and symmetrical
- air bronchograms may be evident
- hyperinflation (in a non ventilated patient) excludes the diagnosis
- radiographs may show hyperinflation if the patient is intubated.
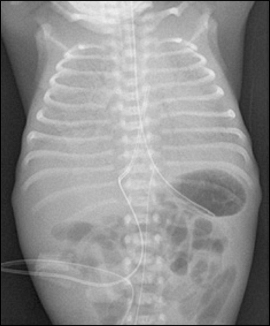 Figure 2: CXR showing 'ground glass' appearance and air bronchogram (www.aafp.org)
Figure 2: CXR showing 'ground glass' appearance and air bronchogram (www.aafp.org)
Full blood count and blood culture
- A full blood count and blood culture should be performed prior to starting antibiotics, but antibiotics should not be withheld if blood sampling proves technically difficult. Antibiotics may be given intramuscularly in these circumstances.
Management
Non-respiratory management of RDS
- Temperature control is an important facet of the care of the infant with respiratory distress and both hypothermia and hyperthermia should be avoided. The temperature should be maintained in the neutral thermal range.
- Enteral feeding should generally be avoided in infants with significant respiratory distress (oxygen requirements greater than 35 per cent). During the initial stabilisation intravenous fluid therapy is useful if it can be started easily but premature infants do not necessarily require an IV immediately. Exposure to manipulation and cold stress may do more harm than good in this situation. Attention to prevention of hypoglycaemia is, however, an important part of ongoing care.
- Minimal handling is important and can be facilitated by the use of monitors to help assess the infant's status (cardiorespiratory, temperature and oxygen saturation monitors should be used whenever possible).
- Antibiotics - commence penicillin and gentamicin therapy after initial investigations.
Respiratory management of RDS
Airway
Placing the unwell infant in the prone position rather than supine may provide a clear airway. Ensure the infant has full cardiorespiratory and saturation monitoring when placing prone. Repeated suctioning of the pharynx is not required and may cause apnoea and hypoxia.
Oxygen
Both too much and too little oxygen are bad for preterm infants, hypoxia is much more dangerous over the short period of time while awaiting transport.
Monitoring of oxygen
- Capillary acid base status (CAB) allows monitoring of pCO2 and pH. High pCO2>70 is an indication for intubation
- Arterial blood gases (ABG) - accurate assessments can be made from samples taken from indwelling arterial lines which are usually performed in an intensive care setting, aiming to keep pO2 between 50 and 80 mm Hg. Assessment of oxygen requirements from arterial ‘stabs’ is not reliable.
- Non-invasive monitoring - oxygen saturation monitors may be attached to the infant’s right hand (pre-ductal). The desired range for infants is 91 - 95 percent.
- Cyanotic threshold - keep the ambient oxygen concentration 5 to 10 percent above the level at which the infant is noted to be cyanosed, if saturation monitoring is unavailable.
Administration of oxygen
- Oxygen concentrations up to 40 per cent may be achieved through the oxygen port into an incubator.
- Above 40 per cent is achieved using warmed, humidified gas delivered via a headbox if CPAP is not available. If using a headbox, flow rates of at least 8-10 l/min are required to avoid rebreating of carbon dioxide.
- Nasal CPAP.
Non-invasive ventilation – Nasal CPAP
CPAP will prevent collapse of alveoli, recruit more alveoli and help to increase the functional residual capacity (FRC) thus helping in better oxygenation. It is effective in reducing the need for intubation.
Indications for NCPAP in babies having continuing care in Level 4-5 Neonatal Units
- Birth weight >1499g and gestation greater than 32 weeks.
- Less than 24 hours old (the use of rescue NCPAP in a 2-3 day old baby with progressive respiratory failure is often followed by the need for rescue endotracheal intubation and aggressive mechanical ventilation).
- Have clinical signs of respiratory distress.
- Require FiO2 of at least >0.25 to maintain a saturation between 91-95%.
- A chest radiograph consistent with mild respiratory distress syndrome or transient tachypnoea of the newborn.
Contraindications to NCPAP for ongoing management in Level 4-5 Neonatal Units
- Birth weight < 1500g , gestation <32 weeks
- More than 24 hours old at initiation of NCPAP
- Persistent FiO2 >0.40 (after commencing NCPAP)
- Persistent hypercarbia (PaCO2 >60mmHg) with respiratory acidosis (pH<7.25)
- Apnoea (babies >1499g and greater than 32 weeks gestation rarely have uncomplicated apnoea of prematurity as a reason to require NCPAP)
- Babies who remain dependant on NCPAP for >72 hours
- Babies with meconium aspiration syndrome, pneumonia, or with a history of a significant asphyxial event. These babies can rapidly develop severe physiological instability with accompanying increased mortality risk.
Intubation and intermittent positive pressure ventilation (IPPV)
Indications
- FiO2>40%
- severe recurrent apnoea
- respiratory failure (pCO2 > 70 and pH < 7.2)
See Intubation procedure for further details.
Ventilator settings for stabilisation
For an infant with RDS reasonable settings are:
- rate 40-60 breaths per minute
- inspiratory time 0.3 sec (expiratory time 0.7 sec)
- PIP (Peak inspiratory pressure) () should be set to achieve ‘reasonable’ chest movement. Usual initial settings in RDS will lie between 20 and 30 cm water
- PEEP 5 cm
- Oxygen set to maintain saturations of 91- 95%.
Surfactant administration
Surfactant administration should be considered in any premature intubated infant with a presumed diagnosis of RDS.
Sudden deterioration
Spontaneously ventilating infant
In a spontaneously ventilating infant sudden deterioration may be caused by:
- pneumothorax
- failure of oxygen supply
- increase in severity of the underlying disease
- persistent pulmonary hypertension (PPH)
Infants may be able to sustain the protracted tachypnoea associated with RDS for hours or days before developing respiratory failure. This may be signalled by onset of apnoea, sudden increase in oxygen requirements or laboured respiratory efforts.
Continued close monitoring throughout the infant's entire illness is mandatory.
Ventilated infant
In a ventilated infant sudden deterioration is most likely to be caused by:
- pneumothorax this is easily overlooked clinically so transillumination or a further chest x-ray are essential
- endotracheal tube blockage or displacement
- mechanical failure with the ventilator
- increase in the severity of the underlying lung disease
- massive intraventricular haemorrhage
- necrotising enterocolitis, especially if perforation has occurred
- patent ductus arteriosus
More information
References
- Stabilisation and transport of newborn infants and at-risk pregnancies 4th Edition 1998 NETS Publication
- Neonatal-perinatal medicine: Diseases of the fetus and infant 6th ed 1997. Fanaroff AA and Martin RJ (eds). Mosby-Year Book.
Further reading
- The following reviews published in the Cochrane Library provide more detail on the evidence underlying use of exogenous surfactant in RDS, choice of exogenous surfactant, timing of surfactant therapy and choice of ventilator rate.
- Greenough A, Milner AD, Dimitriou G. Synchronised mechanical ventilation for respiratory support in newborn infants (Cochrane Review). In: The Cochrane Library, Issue 2, 2001. Oxford: Update Software.
- Soll RF. Synthetic surfactant for respiratory distress syndrome in preterm infants (Cochrane Review). In: The Cochrane Library, Issue 2, 2001. Oxford: Update Software.
- Soll RF, Blanco F. Natural surfactant extract versus synthetic surfactant for neonatal respiratory distress syndrome (Cochrane Review). In: The Cochrane Library, Issue 2, 2001. Oxford: Update Software.
- Yost CC, Soll RF. Early versus delayed selective surfactant treatment for neonatal respiratory distress syndrome (Cochrane Review). In: The Cochrane Library, Issue 2, 2001. Oxford: Update Software.
- Photo Gallery Index: Newborn Nursery at LPCH - Stanford University, School of Medicine
- Respiratory Distress in the Newborn - American Family Physician
Get in touch
Version history
First published: February 2015
Review by: February 2018

