There are mandatory timelines around sentinel events reporting, reviews and recommendations.
1. Access the portal
If you (or your health service) don't have access to the sentinel events portal, please complete the onboarding form and submit to SCV.
- The onboarding form must be accompanied by written approval from a relevant director or executive from the health service.
- All onboarding requests are reviewed by the sentinel events team to ensure they meet the access requirements.
- Once approved access should be granted within 24-48 hours.
Read about the sentinel events portal.
What's new and how to get access
You can also read our FAQS or contact us if you have any questions.
2. Submit the sentinel event
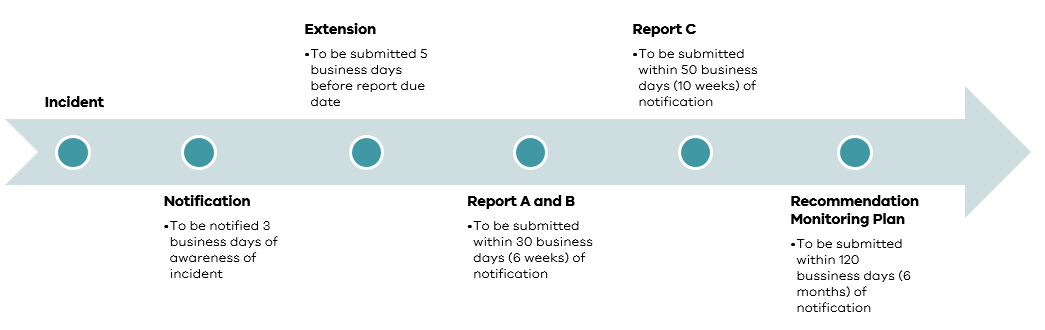
Notify us within three business days of becoming aware of a sentinel event.
Notify a sentinel event
Unsure if you need to report?
Read the list of sentinel event categories. Or contact us.
Does the event involve multiple health services?
The health service that provided the final care related to the adverse event should be responsible for notifying the event, initiating the review and engaging the other health services. This can be negotiated on a case by case basis, SCV can provide advice if required.
All services involved in the care of the patient should participate in the review.
3. Conduct a review
Form a review panel that includes:
To avoid delays, check the availability of team members for the duration of the review.
Involve patients and their families in the review
Please consider the patients, their families, carers and their friends during the review process.
Families can provide crucial and insightful information during the review of a serious adverse event.
Submit your review report
Within 30 business days (six weeks) of notifying us, complete and submit the part A and B report template.
This includes the"
- description of the event
- timeline of the event
- analysis of the event
- findings from the review.
Need an extension?
Submit an extension request through the portal at least five business days before parts A and B are due.
You can request a maximum extension of four weeks.
4. Make recommendations
Within 50 business days (10 weeks) of notifying us, complete and submit part C of the sentinel event report.
This includes the recommendations developed from the findings that were determined.
5. Implement your recommendations
Within 120 business days of notifying us, complete and submit the part D, the recommendation monitoring report.
This includes:
- the recommendations
- the actions required to implement the recommendations
- the status of the recommendation
- date recommendation implemented.
For recommendations that remain open 12 months post notification a second part D recommendation monitoring report is required to be submitted.



